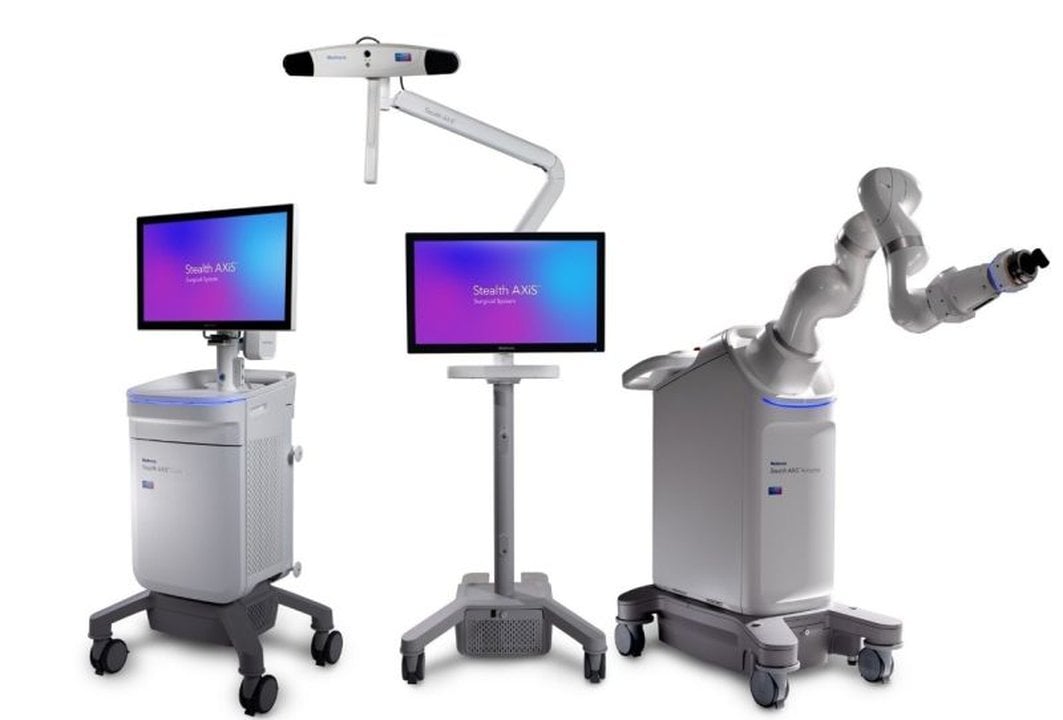
Medtronic Cranial and Spine Therapies has achieved a significant milestone with the CE Mark approval of its Stealth AXiS Surgical System. This endorsement marks an important step towards making advanced technology available to clinicians throughout Europe.
The Stealth AXiS Surgical System is designed to support high‑precision, image‑guided procedures for both spine and cranial surgeries. Earlier this year, the system also received clearance from the US Food and Drug Administration, highlighting the technology’s maturity and its readiness for spinal interventions.
KUKA, a key partner in this development, expressed pride in collaborating with Medtronic on this innovative journey. The partnership reflects how close cooperation can produce tangible benefits in medical settings, improving outcomes in the operating theatre.
This latest approval signifies a major advancement for Medtronic, building on its strong reputation in the medical device industry. The new system is set to enhance precision in surgical procedures, potentially improving patient outcomes across Europe.
View more news from KUKA